Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Muroya, Yusa*; Lin, M.; De Waele, V.*; Hatano, Yoshihiko; Katsumura, Yosuke; Mostafavi, M.*
Journal of Physical Chemistry Letters (Internet), 1(1), p.331 - 335, 2010/01
Times Cited Count:43 Percentile:81.54(Chemistry, Physical)For the first time, a set-up is performed to carry out picoseconds time-resolved experiments in Supercritical Water (SCW) by pulse radiolysis. The kinetics of the decay of the hydrated electron is measured by transient absorption in the near IR on a time-scale ranging from 50 ps to 6 ns. The decay of the hydrated electrons in supercritical conditions shows a fast component at a short time below 500 ps, depending on the density of SCW. The results show that in SCW the main reaction for the decay of hydrated electron is that with H O
O .
.
Lin, M.; Fu, H.*; Lampre, I.*; De Waele, V.*; Muroya, Yusa*; Yan, Y.*; Yamashita, Shinichi; Katsumura, Yosuke; Mostafavi, M.*
Journal of Physical Chemistry A, 113(44), p.12193 - 12198, 2009/10
Times Cited Count:7 Percentile:22.38(Chemistry, Physical)With a revisit of the absorption coefficient of the solvated electron in propane-1,2,3-triol, the temperature dependent behavior of the absorption spectrum of solvated electron was studied from room temperature to 573 K by pulse radiolysis techniques. The change in the absorption spectrum of solvated electron in propane-1,2,3-triol observed by cooling down from a high temperature to 333 K is compared with that occurring during the electron solvation process at 333 K. The effect of the specific molecular structure of propane-1,2,3-triol compared to other alcohols is discussed.
Lin, M.; Kumagai, Yuta; Lampre, I.*; Coudert, F.*; Muroya, Yusa*; Boutin, A.*; Mostafavi, M.*; Katsumura, Yosuke
Journal of Physical Chemistry A, 111(18), p.3548 - 3553, 2007/05
Times Cited Count:9 Percentile:30.82(Chemistry, Physical)Lin, M.; Mostafavi, M.*; Muroya, Yusa*; Lampre, I.*; Katsumura, Yosuke
Nuclear Science and Techniques, 18(1), p.2 - 9, 2007/02
Times Cited Count:1 Percentile:14.16(Nuclear Science & Technology)Lin, M.; Kumagai, Yuta; Muroya, Yusa*; Katsumura, Yosuke; Lampre, I.*; Coudert, F.*; Boutin, A.*; Mostafavi, M.*
no journal, ,
In this work, we measure the absorption spectra of hydrated electron in D O solutions containing different concentrations of Li
O solutions containing different concentrations of Li and Mg
and Mg cations at various temperatures by pulse radiolysis techniques, and perform quantum classical molecular dynamics (QCMD) simulations to explain the shift of the absorption spectra due to the combined effects of temperature and salt concentration.
cations at various temperatures by pulse radiolysis techniques, and perform quantum classical molecular dynamics (QCMD) simulations to explain the shift of the absorption spectra due to the combined effects of temperature and salt concentration.
Muroya, Yusa*; Lin, M.; Han, Z.*; Kumagai, Yuta; Lampre, I.*; Mostafavi, M.*; Katsumura, Yosuke
no journal, ,
In this work, we have studied the yield of solvated electron in ethylene glycol, 1,2-propanediol and 1,3-propanediol by electron beam radiation. Their time dependent behaviors in picosecond to microsecond range were also studied by a combination of picosecond and nanosecond pulse radiolysis techniques.

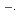 and Br
and Br
 in aqueous solutions
in aqueous solutionsLin, M.; Katsumura, Yosuke; Muroya, Yusa*; Fu, H.*; Yan, Y.*; Yamashita, Shinichi; Mostafavi, M.*
no journal, ,
The absorption spectra of Br
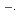 and Br
and Br
 in aqueous solutions are investigated by pulse radiolysis techniques from room temperature to 380 and 350
in aqueous solutions are investigated by pulse radiolysis techniques from room temperature to 380 and 350  C, respectively. Temperature dependent spectrum of Br
C, respectively. Temperature dependent spectrum of Br is also measured under steady-sate. If the absorption of Br
is also measured under steady-sate. If the absorption of Br is of Charge Transfer To Solvent (CTTS), those of Br
is of Charge Transfer To Solvent (CTTS), those of Br
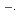 and Br
and Br
 are not.
are not.
Muroya, Yusa*; Lin, M.; Han, Z.*; Yamashita, Shinichi; Ueda, Toru*; Hatano, Yoshihiko; Mostafavi, M.*; Katsumura, Yosuke
no journal, ,
By using a laser-driven S-band linear accelerator and a femtosecond laser, a new highly time-resolved pulse radiolysis system was developed for investigating radiation-induced fast phenomena in high temperature / high pressure water. Transient absorption spectra and fast kinetics of hydrated electron within several nanoseconds at elevated temperatures from room temperature up to supercritical state (400 C, 40 MPa) have been measured.
C, 40 MPa) have been measured.
Muroya, Yusa*; Lin, M.; Yan, Y.*; Han, Z.*; Yamashita, Shinichi; Ueda, Toru*; Mostafavi, M.*; Katsumura, Yosuke*
no journal, ,
no abstracts in English